Everyone shines in their own way.
Wang, Hui
王晖
Greater Bay Biomedical InnoCenter
Junior Principal Investigator
hwang@szbl.ac.cn
Timeline
-
2021 - Present
Shenzhen Bay Laboratory Junior Principal Investigator
-
2015 - 2021
HHMI and University of Washington Research Specialist
-
2009 - 2015
HHMI and University of Washington Postdoc Fellow
-
2008 - 2009
Nankai University Research Associate
-
2002 - 2008
Tsinghua University Ph.D.
-
1998 - 2002
Sichuan University B.S.
Research Areas
Dr. Hui Wang studies iron biology and how this essential element is implicated in a variety of critical processes, such as iron metabolism, iron homeostasis, and ferroptosis. Her group, by using a combination of interdisciplinary approaches, seeks to understand the key iron-involving regulatory mechanisms by which cells maintain their homeostasis in response to signals or stresses, to uncover the iron-relevant pathways that are altered and misregulated in diseases, and to further explore therapeutical designs to benefit human health based on the learned biological principles.
Highlights
Dr. Hui Wang obtained her Ph.D. degree in Biology from Tsinghua University under the supervision of Prof. Zihe Rao. Shortly after that she joined the HHMI lab led by Prof. Ning Zheng at University of Washington as a postdoctoral researcher, and stayed at HHMI as a research specialist thereafter. Dr. Wang has long been focused on probing the fundamental regulatory mechanisms underlying celluar homeostasis, especially iron homeostasis, to ultimately understand the intricate relationship between the altered/misregulated metabolic pathways and human diseases.
Dr. Wang discovered for the first time that an FeS cluster functions as a critical redox switch in the ubiquitin-proteasome system in eukaryotes. In response to the changes of oxygen/redox levels in cells, this cluster switches between its reduced and oxidized states, dictating the degradation of IRP2, an essential controller in iron metabolism, and thereby the maintainance of iron homeostasis. Her findings solidified the role of FeS proteins in sensing of oxygen/redox to determine the critical processes. Additionally, Dr. Wang has explored the RNA metabolism, and published first-authored articles in Molecular Cell, Nature Structural & Molecular Biology, Cell Research, The EMBO Journal, and Journal of Molecular Biology. Her work has been previewed and highlighted at several occasions, and is well-recognized within the community.
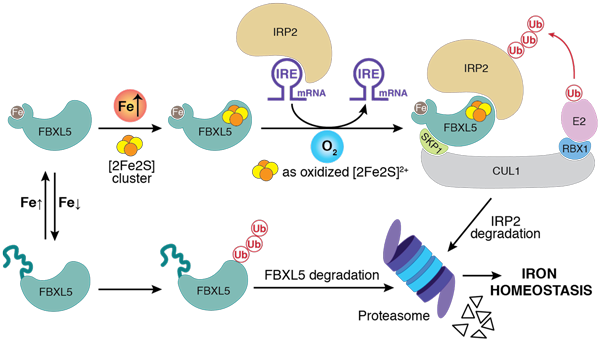
Mol Cell. 2020, 78(1): 31-41.
Dr. Wang unveiled a novel oxygen/redox sensing regulatory mechanism to maintain iron homeostasis
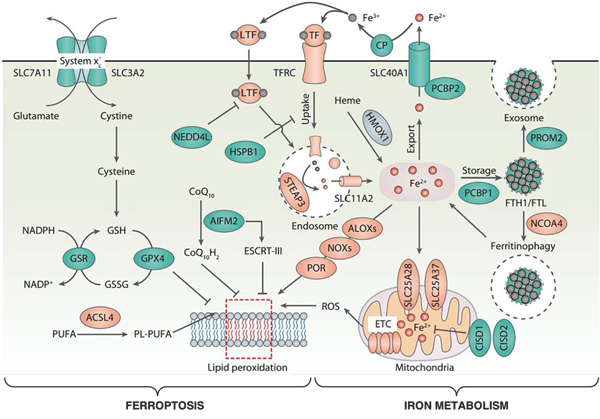
Front Cell Dev Biol. 2020, Oct 7;8:590226.
Dr. Wang’s group strives to understand the interplay between iron metabolism and ferroptosis
Honors
• 2019 10 years of Dedicated Service HHMI
• 2005 General Electric Scholarship Tsinghua University
• 2004 Guanghua Scholarship Tsinghua University
• 2002 Outstanding Graduate Cadre Sichuan University
• 2002 Dikang Scholarship Sichuan University
Related News
Preview Mol Cell. 2020, 78(1): 1-3, https://doi.org/10.1016/j.molcel.2020.03.020
Research Highlight Nat Chem Biol. 2020, 16(5): 481, https://doi.org/10.1038/s41589-020-0536-7
Editors’ Choice Sci Signal. 2019, 12(588): eaay5593, https://doi.org/10.1126/scisignal.aay5593
News & Views EMBO J. 2020, 39(8): e104744, https://doi.org/10.15252/embj.2020104744
Selected Publications
1. Wang H*, Shi H*, Rajan M, Canarie RE, Hong S, Simoneschi D, Pagano M, Bush FM, Stoll S, Leibold AE, Zheng N. FBXL5 regulates IRP2 stability in iron homeostasis via an oxygen-responsive [2Fe2S] cluster. Mol Cell. 2020, 78(1): 31-41.
2. Kuchay S*, Wang H*, Marzio A, Jain K, Homer H, Fehrenbacher N, Philips MR, Zheng N, Pagano M. GGTase3 is a newly identified geranylgeranyltransferase targeting a ubiquitin ligase. Nat Struct Mol Biol. 2019, 26(7): 628-636.
3. Wang H, Curran EC, Hinds TR, Wang EH, Zheng N. Crystal structure of a TAF1-TAF7 complex in human transcription factor IID reveals a promoter binding module. Cell Res. 2014, 24(12): 1433-1444.
4. Wang H, Morita M, Yang X, Suzuki T, Yang W, Wang J, Ito K, Wang Q, Zhao C, Bartlam M, Yamamoto T, Rao Z. Crystal structure of the human CNOT6L nuclease domain reveals strict poly(A) substrate specificity. EMBO J. 2010, 29(15): 2566-2576.
5. Wang H*, Pang H*, Bartlam M, Rao Z. Crystal structure of human E1 enzyme and its complex with a substrate analog reveals the mechanism of its phosphatase/enolase activity. J Mol Biol. 2005, 348(4): 917-926.